Deleted
Deleted Member
Posts: 0
|
Post by Deleted on Oct 27, 2020 10:32:01 GMT 10
It's an interesting read to go back to page 1 of this post to see what we were thinking as this virus emerged.
Anyway, time to reach for the top shelf...does whisky go with donuts?
|
|
|
|
Post by caskur on Oct 27, 2020 12:24:47 GMT 10
LOL... Victoria freed up slightly...
WA people can go to tropical hotspots like Christmas Island and Cocos Islands.
|
|
|
|
Post by caskur on Oct 27, 2020 13:17:21 GMT 10
I just listened to Albo's speech on Melbourne lifting restrictions... EXCELLENT speech...
Now I'm listening to that PM wanker...
Hopefully next election he is gone... He's a lying headcase that bloke is... He needs flogging for his hypocrisy.
GET RID OF THE LIBS.
|
|
|
|
Post by Gort on Oct 27, 2020 14:17:39 GMT 10
I just listened to Albo's speech on Melbourne lifting restrictions... EXCELLENT speech... Now I'm listening to that PM wanker... Hopefully next election he is gone... He's a lying headcase that bloke is... He needs flogging for his hypocrisy. GET RID OF THE LIBS. Poor old Albo, that whiny voice of his ... such a turn-off.   |
|
|
|
Post by caskur on Oct 27, 2020 15:18:42 GMT 10
I don't care. His speech was from an honest genuine guy.... it's a bit nasally..
But Morrison is a wanker... He'll be out next time. He stood up praising Andrews and yet these past months have told everyone to have open borders... The guy is a fundy lunatic.
I want him OUT.
Andrews killed it being the dictator...
The hurdle now is to make sure the basics are kept up. Social distance, masks, testing, washing hands,... obeying curfews, whatever it takes.
|
|
|
|
Post by Gort on Nov 10, 2020 9:19:58 GMT 10
So, it seems that there is light at the end of the tunnel. A vaccine with 90% effectiveness could be on the way "fairly soon". This is good news.  |
|
|
|
Post by Gort on Nov 10, 2020 10:46:45 GMT 10
Pfizer says COVID-19 vaccine could be 90 per cent effective The shots made by Pfizer and its German partner BioNTech are among 10 possible vaccine candidates in late-stage testing around the world.(Pexels: Polina Tankilevitch) A COVID-19 vaccine being produced by Pfizer may be 90 per cent effective in stopping the transmission of the deadly virus. Early, but incomplete, test results from the drug manufacturer have showed promising results in 94 infections being treated in the trial. The news has brought a burst of optimism to a world desperate to bring the catastrophic outbreak under control. The Pfizer trial is one of four potential vaccines the Australian Government has signed an agreement to purchase if it is successful. Federal Health Minister Greg Hunt said the Government would continue to monitor further results as they are made available. "The data on our vaccine candidates continues to be positive. We will examine the evidence carefully but the latest results are heartening news," he said. Pfizer, which is developing the vaccine with its German partner BioNTech, is on track to apply for emergency use approval from the US Food and Drug Administration (FDA).
Even if all goes well, authorities have stressed it is unlikely any vaccine will arrive before the end of the year.Pfizer's senior vice-president of clinical development, Bill Gruber, told the Associated Press the company was "very encouraged" by the early results. Results are promising but more needs to be done The announcement doesn't mean for certain that a vaccine is imminent. This interim analysis, from an independent data monitoring board, looked at 94 infections recorded so far in a study that has enrolled nearly 44,000 people in six countries. Some participants got the vaccine, while others received placebos. Pfizer have not released specific breakdowns for the results. The study is continuing and Pfizer cautioned that the protection rate might change as more COVID-19 cases are added to the calculations. Participants were tested only if they developed symptoms, leaving unanswered whether vaccinated people could get infected but show no symptoms and unknowingly spread the virus. The FDA has required US vaccine candidates be studied in at least 30,000 people. The study must include an adequate numbers of older adults, and people from high risk groups including minorities and people with chronic health problems. Pfizer doesn't plan to stop its study until it records 164 infections among all the volunteers, a number that the FDA has agreed is enough to tell how well the vaccine is working. The agency has made clear any vaccine must be at least 50 per cent effective. Vaccine optimism boosted by early results Dr Anthony Fauci, the US Government's top infectious disease expert, said the results suggesting 90 per cent effectiveness were "just extraordinary". "Not very many people expected it would be as high as that," he said. "It's going to have a major impact on everything we do with respect to COVID." Dr Bruce Aylward, the World Health Organization's senior adviser, said Pfizer's vaccine could "fundamentally change the direction of this crisis" by March, when the UN agency hopes to start vaccinating high risk groups.Global markets, already buoyed by the victory of president-elect Joe Biden, rocketed on the news from Pfizer. The S&P 500 was up 3.3 per cent in afternoon trading, while the Dow Jones gained more than 1,300 points. Pfizer jumped more than 11 percent and other vaccine stocks were up as well. Pfizer has estimated it could have 50 million doses — enough for 25 million people — available globally by the end of 2020.www.abc.net.au/news/2020-11-10/pfizer-says-vaccine-data-suggests-may-be-90-per-cent-effective/12865626
|
|
|
|
Post by Gort on Nov 10, 2020 10:48:42 GMT 10
Hopes rise for end of pandemic as Pfizer says vaccine is 90% effectiveGlobal stocks surge and experts optimistic as Covid vaccine exceeds expectations 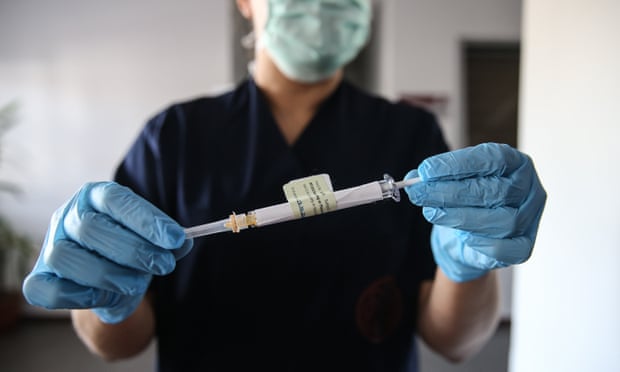 Hopes are soaring that a Covid vaccine is within reach, following news that an interim analysis has shown Pfizer/BioNTech’s candidate was 90% effective in protecting people from transmission of the virus in global trials. Hopes are soaring that a Covid vaccine is within reach, following news that an interim analysis has shown Pfizer/BioNTech’s candidate was 90% effective in protecting people from transmission of the virus in global trials.The vaccine performed much better than most experts had hoped for, according to the companies’ analysis, and brings into view a potential end to a pandemic that has killed more than a million people, battered economies and upended daily life worldwide. The data is from an interim analysis and the trial continues into December but the headline results were emphatic. Regulators will be looking to process an emergency licence application at record speed. Global stocks surged to record highs on the news, while companies that have become synonymous with lockdowns in the west, including Zoom’s video conferencing and food delivery firms, saw falls in their value. John Bell, Regius professor of medicine at Oxford University, who is involved with the Oxford vaccine, said that the Pfizer team had shown “an amazing level of efficacy” and it could mean a return to normality by spring. “I’m the first guy to say that but I will say it with some confidence,” he told the BBC.Manufacturing is already under way. Pfizer said they expect to supply globally up to 50m vaccine doses in 2020 and up to 1.3bn doses in 2021. Countries will decide who they prioritise for vaccination. In the UK, the joint committee on vaccination and immunisation has recommended that – presuming the vaccines work well enough in elderly people – the first vaccines go to care home workers and residents, followed by anybody over 80 and other health and social care workers. After that, people are expected to get them in decreasing age order.The news comes too late to help Donald Trump’s re-election campaign in the US, but the vice-president, Mike Pence, tried to claim their administration’s Operation Warp Speed programme had helped the vaccine’s development. Pfizer denied the suggestion. “We were never part of the Warp Speed,” Kathrin Jansen, a senior vice-president and the head of vaccine research and development at Pfizer, said in an interview. “We have never taken any money from the US government, or from anyone.” BioNTech, the small biotechnology company that is the originator of the vaccine, was founded by two married German scientists, Uğur Şahin and Özlem Türeci, both born to Turkish immigrant parents, and the Austrian oncologist Christopher Huber. It originally set out to develop new types of immunotherapy for cancer, but has concentrated its capacities on the race for a Covid-19 vaccine. There are so far no safety concerns around the vaccine, with the two companies reporting no serious side-effects. The high percentage of those protected makes it especially compelling. Regulators have previously said they would approve a vaccine that has just a 50% effectiveness rate – protecting half of those who get vaccinated. “Today is a great day for science and humanity. The first set of results from our phase 3 Covid-19 vaccine trial provides the initial evidence of our vaccine’s ability to prevent Covid-19,” said Dr Albert Bourla, the Pfizer chairman and chief executive. “We are reaching this critical milestone in our vaccine development programme at a time when the world needs it most with infection rates setting new records, hospitals nearing over-capacity and economies struggling to reopen.” Other scientists tried to marry caution with their obvious excitement at the early results, warning that the full data was not yet available and that the trial was continuing. “In the absence of any data from Pfizer and BioNTech, we have to take these very exciting claims at face value,” said Dr Simon Clarke, an associate professor of cellular microbiology at the University of Reading. “It seems highly unlikely that a major pharmaceutical company would get such eagerly awaited news wrong.” Prof Peter Horby of Oxford University, who runs the Recovery Covid drug trial, said it seemed like an important moment. “This news made me smile from ear to ear,” he said. “It is a relief to see such positive results on this vaccine and bodes well for Covid-19 vaccines in general. Of course we need to see more detail and await the final results, and there is a long, long way to go before vaccines will start to make a real difference, but this feels to me like a watershed moment.” Pfizer/BioNTech’s mRNA vaccine, which uses the genetic code rather than any part of the virus itself, has been one of the leaders in the global vaccine race. Interim results are also expected this month or in early December from trials for the Oxford University/AstraZeneca vaccine, which is based on a different technology. The phase 3 trials have involved more than 43,000 people and are intended to establish whether the vaccine works. Volunteers are given either the Covid vaccine, which was administered in two shots about three weeks apart, or a placebo alternative such as the meningitis vaccine, and neither they nor their doctors know which they have had. There have been concerns that black and minority ethnic populations are particularly vulnerable to Covid. Approximately 42% of global participants and 30% of US participants in the trials have racially and ethnically diverse backgrounds and appear to have been as well-protected as everyone else, the company says. But one important unanswered question is how long the vaccine will last. The US and UK both welcomed the development but urged patience. “I congratulate the brilliant women and men who helped produce this breakthrough and to give us so much cause for hope,” the US president-elect, Joe Biden, said. “It is also important to understand that the end of the battle against Covid-19 is still months away … a mask remains a more potent weapon against the virus than a vaccine. Today’s news does not change this urgent reality.” The UK’s prime minister and deputy medical adviser warned that nobody should assume the pandemic was over and let down their guard. “We can’t let our enthusiasm run away with us,” said Boris Johnson, adding that the public must continue to observe the lockdown restrictions. Prof Jonathan Van-Tam said the results showed the vaccine prevented disease, but “we don’t know what this means yet for when we can get life back to normal … please don’t relax.” Many countries already have orders for the Pfizer/BioNTech vaccine. The UK has bought 40m doses – enough for 20 million people because two doses are required. The EU has secured 200m doses, which it will distribute. The companies have a $1.95bn (£1.5bn) contract with the US government to deliver 100m vaccine doses beginning this year. www.theguardian.com/world/2020/nov/09/covid-19-vaccine-candidate-effective-pfizer-biontech
|
|
|
|
Post by Gort on Nov 13, 2020 9:54:29 GMT 10
Oh FFS ...  Passenger Aboard First Cruise Ship to Return to Sailing in Caribbean Tests Positive for COVID-19 Passenger Aboard First Cruise Ship to Return to Sailing in Caribbean Tests Positive for COVID-19Passengers and non-essential crew are now isolating in their cabins as the ship heads back toward its departure point in Barbados November 11, 2020 05:39 PM The first cruise ship to set sail in the Caribbean since the beginning of the COVID-19 pandemic has reported that a passenger on board has tested positive for the virus, according to The Points Guy. A reporter for the travel site, Gene Sloan, is aboard the SeaDream 1 and was meant to be documenting the new safety measures on the small vessel, which departed from Barbados on Saturday and is carrying 53 passengers and 66 crew. Sloan reported that the boat's captain, Torbjorn Lund, made an announcement over the intercom midday on Wednesday explaining that a passenger had tested positive "on a preliminary basis." It's currently unclear what he means by "preliminary." Sea Dream Yacht Club, which owns the ship, did not immediately reply to PEOPLE's request for clarification.  SeaDream 1 at a port in Norway in August 2020 | CREDIT: SONDRE SKJELVIK / GETTY Lund reportedly said in his announcement that the crew was working under the assumption that they had at least one COVID patient on board and noted that the passenger who tested positive had been feeling ill before the test. Sloan detailed that the captain asked all passengers and non-essential crew to immediately return to their cabins to isolate. The doctors on board then began systematically testing passengers using three rapid COVID testing machines. Everyone on the ship had already been tested several days before departing, the day of their departure, and again several days into the trip. While the boat made several island stops already, passengers were only allowed to visit empty beaches and did not come into contact with locals as part of the effort to protect communities from potential infection. SeaDream 1 is now heading back to the port from which it departed, where it will be met by medical professionals. Cruise companies halted all sailings in the Caribbean last spring as the pandemic took hold in North America, individual countries enacted stringent travel restrictions and the U.S. Centers for Disease Control and Prevention (CDC) enacted a "no-sail order." "This is a blow to the cruise industry's efforts to restart operations in the Caribbean," Sloan told PEOPLE. "SeaDream's return to cruising in the Caribbean was a watershed moment for the industry, and many were hoping it would go smoothly." The CDC first issued a no-sail order on March 14 and was intended to stay in place for 30 days. At the time, several cruise ships across the world had become sources of major coronavirus outbreaks. One ship, the Grand Princess, was quarantined off San Francisco after 21 people on board tested positive for the virus in March. That ship eventually docked in the port of Oakland and those on board quarantined on land. Another, the Holland America, reported four dead and 233 ill on two of its ships heading for Ft. Lauderdale in March after being turned away from ports in South America. In October, the CDC announced it would lift its no-sail order as of November 1 after 8 months, with a set of strict new health and safety protocols for cruise operators. The agency is also requiring all ships that pass through U.S. waters and can carry more than 250 passengers to run mock voyages with volunteer passengers and crew on board in order to "test cruise ship operators' ability to mitigate COVID-19 risk." (The SeaDream 1 only carries 112.) Cruise lines including Royal Caribbean announced just this week that they were looking for volunteer passengers to join such sailings, according to Cruise Industry News. "This framework provides a pathway to resume safe and responsible sailing. It will mitigate the risk of COVID-19 outbreaks on ships and prevent passengers and crew from seeding outbreaks at ports and in the communities where they live," CDC Director Robert R. Redfield, M.D said in a press release. "CDC and the cruise industry have a shared goal to protect crew, passengers, and communities and will continue to work together to ensure that all necessary public health procedures are in place before cruise ships begin sailing with passengers." "The $150 billion cruise industry has been working around-the-clock for months to find safe ways to restart sailings," says Sloan. "This small-ship voyage was being closely watched by other cruise lines, cruise fans and the industry's regulators, and — depending on what happens — this could be yet another disappointing setback for the industry and the millions of vacationers who are looking to return to the sea." people.com/travel/passenger-aboard-first-cruise-ship-to-return-to-sailing-in-caribbean-tests-positive-for-covid-19/ |
|
|
|
Post by Gort on Nov 13, 2020 12:46:31 GMT 10
Government details COVID-19 vaccination programBy Rachel Clun People may have to show proof they’ve received a COVID-19 vaccination if they wish to enter Australia according to the federal government’s COVID-19 vaccination policy. Any COVID-19 vaccine will be free for all Australians or medicare-eligible visa holders. “While the Australian Government strongly supports immunisation and will run a strong campaign to encourage vaccination, it is not mandatory and individuals may choose not to vaccinate,” the Australian COVID-19 Vaccination Policy said. “There may however, be circumstances where the Australian Government and other governments may introduce border entry or re-entry requirements that are conditional on proof of vaccination.” The first roll-out of the country’s COVID-19 vaccinations program will not fall under the national immunisation program, which is used for vaccinations including measles and influenza. Elderly, vulnerable front of the line in national COVID vaccination policyBy Marissa Calligeros The National Cabinet has also agreed to a national COVID vaccination policy, which will prioritise the elderly and people particularly vulnerable to the virus. "Those that are caring for vulnerable people, vulnerable people themselves and those at highest risk of transmission will be at the highest priority," Acting Chief Medical Officer Paul Kelly said.  Prime Minister Scott Morrison is joined by Chief Medical Officer Profesor Paul Kelly (middle) and Professor Alan Finkel (back) for a press conference on Friday.CREDIT:ALEX ELLINGHAUSEN He said the federal Department of Health will house a new vaccine division and states and territories would continue to be "very engaged" in the distribution of a COVID vaccine. "The final point was made very clearly by the PM, as he's done previously, is that this vaccine will be free for all. And all Australians, for anyone who wants to take that vaccine." Professor Kelly said he had no doubt many Australians will be queuing for a COVID vaccine, despite vaccine sceptics. "We are an excellent immunisation nation," he said. "Most people agree with immunisation being an important component of our preventative strategy for ensuring the health of the nation. We roll out new immunisation programs often. "I would say this has particular challenges as to the newness of it and so forth. And so we're very aware that there will be people that are not wanting to have this vaccination or to spread information which is not true." Professor Kelly said communication was a strong part of the government's policy. "That's already starting, putting out information about the types of vaccines, their advantages and so forth. As the PM said, absolutely, there are no short cuts to this. It's going fast, that's true, but all of the processes for regulation will be there. "I'm sure there will be a lot of people that will be queuing up for this vaccine through next year." The government expects the initial supply of any vaccines to be limited, so people in “priority groups” will be vaccinated first. But deciding which groups to make a priority is “difficult and contentious”, the vaccination policy said. So far, three groups have been identified. The first group includes those who are at increased risk of exposure to the virus (and at higher risk of being infected and transmitting) and who work in settings with a high transmission risk. That group includes care workers, including health, aged care, disability support and quarantine workers. The second group includes those at an increased risk of developing severe disease or having worse health outcomes, including Aboriginal and Torres Strait Islander people, the elderly, and people with some underlying health conditions. Essential services workers form the last group, which includes “key occupations” that cover services “critical to societal functioning”. The paper does not explain which occupations that may include. www.theage.com.au/national/coronavirus-live-updates-victoria-chases-13th-day-of-no-new-cases-nsw-seeks-border-opening-usa-europe-continue-to-face-growing-wave-20201113-p56e91.html
|
|
|
|
Post by caskur on Nov 13, 2020 13:27:02 GMT 10
Medical staff + hospital cleaners and blue collar workers should be jabbed first... But there is 25 million in Australia and they were getting more than 25 million jabs so this talk of who gets it first is just pitting groups against each other.
And NO ONE should be allowed to come here from overseas UNLESS you're an Australian citizen.
|
|
|
|
Post by ponto on Nov 13, 2020 13:49:56 GMT 10
Stop the boats and hospital workers first...politicians, Sky and Murdoch news last....preferably not at all while all sent on a cruise to the USA.
|
|
|
|
Post by caskur on Nov 13, 2020 16:34:58 GMT 10
Yep, send them to the USA... that I would like to see.
|
|
|
|
Post by Stellar on Nov 14, 2020 10:27:46 GMT 10
I won't be lining up for a jab! It normally takes 10.7 years to develop a safe and effective vaccine. Any vaccine developed in record time - and I know scientists are throwing everything at this worldwide - does not have the safety issue factored in. We don't know what side effects there will be because that takes time.
I'd be more inclined to wait for the Australian vaccine being developed at the University of Queensland because in initial tests it seems to be more compatible with the elderly.
One thing with vaccines is that they could make the disease worse. It's called "disease enhancement" where those who have been vaccinated become sicker when exposed to the virus than people who have not been vaccinated.
This is just one reason why you need extensive studies with healthy volunteers to pick up potential side-effects. So any vaccine developed over the next 6 months or so still needs years to test to be absolutely certain it is completely safe - or that it works.
|
|
|
|
Post by ponto on Nov 14, 2020 10:55:07 GMT 10
I am going to wait for a jab....see what happens to all the essential services first.
|
|
|
|
Post by caskur on Nov 15, 2020 3:41:16 GMT 10
I won't be lining up for a jab! It normally takes 10.7 years to develop a safe and effective vaccine. Any vaccine developed in record time - and I know scientists are throwing everything at this worldwide - does not have the safety issue factored in. We don't know what side effects there will be because that takes time. I'd be more inclined to wait for the Australian vaccine being developed at the University of Queensland because in initial tests it seems to be more compatible with the elderly. One thing with vaccines is that they could make the disease worse. It's called "disease enhancement" where those who have been vaccinated become sicker when exposed to the virus than people who have not been vaccinated. This is just one reason why you need extensive studies with healthy volunteers to pick up potential side-effects. So any vaccine developed over the next 6 months or so still needs years to test to be absolutely certain it is completely safe - or that it works. An Italian lady friend of mine had a flu shot then developed the worse flu she ever had. I never really trusted vaccines since... I had puppies that I sold vaccinated and two at two different places came down with diseases they were supposed to be vaccinated against. I'm fully vaccinated for everything bar the flu. I haven't had flu for over 7 years. I have a motto... 'stay away from sick people.'  |
|
|
|
Post by Stellar on Nov 15, 2020 9:15:13 GMT 10
That's always a good idea. But what if you don't know they're sick? When someone has the flu it's obvious they're sick and it's obvious we need to keep our distance. However the Covid virus is a serial killer with the ability to travel amongst us undetected and infect almost everyone it comes across. It then gets straight to work doing the only thing it's programmed for and that's reproducing and spreading as far and as fast as it can.
In other words, you need to stay away from all people. Because you don't know who has or doesn't have the virus. If everyone was made to wear masks we'd stop this virus in its tracks. Because then all those people who feel absolutely fine but have the virus unknowingly - the asymptomatic carriers - wouldn't be coughing and sneezing and spreading their droplets where other people would inhale them. And then if you added social distancing and good hand washing, the virus would die out.
But we're not smart enough to do that. It needs at least a minimum of 60% of people to play by the rules. But too many people won't because of their "rights."
And as I've always said, these viruses are much smarter than us so we're always several steps behind playing catch up. They've been described as mindless little scraps of RNA that aren't actually alive, but they're a devious and mortal enemy that can still bring our global society to a crashing halt by causing the greatest public health crisis we've experienced in the past 100 years. And it's best to realise it won't be the last.
|
|
|
|
Post by Gort on Dec 7, 2020 10:12:33 GMT 10
|
|
|
|
Post by Gort on Dec 11, 2020 8:38:06 GMT 10
Too bad about the QLD vaccine candidate ... Who'd have thunk it? Causes a false positive for HIV.  It does NOT give you AIDS. However, they may have been better off if they had had a marketing person involved at the outset!  Marketing guy: Marketing guy: "So ... your vaccine uses a protein from HIV to create a "molecular clamp" for the vaccine to work on COVID-19?" Scientist: "Yep."   Marketing guy: Marketing guy: "Yeah /nah I reckon don't go with that idea."  |
|
|
|
Post by caskur on Dec 11, 2020 11:11:24 GMT 10
Can you believe it?
What were they thinking?
More money down the drain.
|
|
|
|
Post by ponto on Dec 12, 2020 13:25:36 GMT 10
The aussie method of using the recombinant DNA of the aids virus was innovative but required further shots to combat the reaction to the vaccine, the Poms used aids DNA but not recombinant which resulted in just one shot into the arm that as a plus did not need super chilled old storage...more traditional method of making vaccines,
|
|
|
|
Post by pim on Dec 12, 2020 16:27:16 GMT 10
Can you believe it? What were they thinking? More money down the drain. You'd be amazed at the pharmaceutical benefits of quite toxic substances. Talk to a qualified pharmacist about the medicinal properties of spider venom. And snake venom. As antibiotics lose their efficacy because the bugs have developed resistance, all the more reason to get creative and innovative. And to save the rainforests. |
|
|
|
Post by Gort on Dec 12, 2020 16:58:23 GMT 10
As Dr. Evil would say: "It's a start."  US regulators approve Pfizer vaccine, immunisations to start in days US regulators approve Pfizer vaccine, immunisations to start in daysBy Matthew Knott Updated December 12, 2020 — 2.04pmfirst published at 1.28pm Washington: Americans will start receiving coronavirus vaccine shots within days after federal regulators officially granted an emergency use authorisation for Pfizer and BioNTech's vaccine. The much-anticipated approval from the Food and Drug Administration offers a ray of hope after a torrid year in which almost 300,000 Americans have died of COVID-19. The US continues to set daily records for COVID-19 deaths and hospitalisations, with around 3000 people now dying each day from the virus. In a video announcing the approval on Saturday (AEDT), US President Donald Trump said the US had achieved a "medical miracle" and hailed the vaccine as "one of the greatest scientific accomplishments in history". "It will save millions of lives and soon end the pandemic once and for all," Trump said, adding that the first person will receive it in "less than 24 hours". "The pandemic may have begun in China but we are ending it right here in America." Trump said his administration had "given Pfizer and other companies a great deal of money hoping this would be the outcome". Unlike other pharma companies, Pfizer opted not to take money from the US government for research and development into its vaccine. Alex Azar, the head of the Department of Health and Human Services, said earlier in the day: "We could be seeing people getting vaccinated Monday, Tuesday of next week." Azar told Good Morning America that 20 million Americans could be vaccinated in the coming weeks and 50 million by the end of January.  America's vaccine rollout begins this week. Cedars-Sinai Medical Centre in Los Angeles has set up sub-zero storage for the vaccine. CREDIT:CEDARS-SINAI On Friday (AEDT), the FDA's expert advisory panel voted 17-4 to approve the vaccine for Americans aged over 16. The only major disagreement on the panel came over the question of whether the benefits of getting vaccinated outweigh the risks for people aged 16 to 18 given they are less susceptible to the virus. Australia's acting Chief Medical Officer Paul Kelly said the approval of the Pfizer vaccine in the US, UK and Canada will not speed the process in Australia. "The reality is we haven't had a single community case in eight days, we've had very few cases in the community over the last month or so," he said. "We do not have an emergency at the moment so we can afford to wait and go through the processes as we would normally do ... A vaccine is important but next year is when we'll be looking at that." A University of Queensland vaccine trial has been formally abandoned after participants returned false positive HIV results. The Pfizer vaccine, which has been found to be 95 per cent effective, will first be distributed in the US to healthcare workers and nursing home residents before being given to the broader population. The distribution will be an enormous logistical effort given it must be stored at minus 70 degrees Celsius - a far lower temperature than most vaccines. Vaccines usually take years to develop, so the development of an effective coronavirus vaccine in less than a year represents a major scientific achievement. Pharma company Moderna has also applied for emergency use authorisation for its vaccine, which could be approved by the FDA as early as next week. Trump had been growing increasingly impatient with the FDA for not moving quickly enough to approve the Pfizer vaccine. In a tweet on Saturday (AEDT) Trump blasted the FDA as "a big, old, slow turtle" that is "money drenched but heavily bureaucratic". "Get the dam [sic] vaccines out NOW," he instructed the FDA head Stephen Hahn. The vaccine has already been approved in the United Kingdom, Canada, Saudi Arabia and Bahrain. The Washington Post reported that White House chief-of-staff Mark Meadows told Hahn he should prepare to resign unless he approved the vaccine on Saturday (AEDT). Health experts have warned that the US death toll will continue to climb over coming months before the vaccine becomes widely available. Top US infectious diseases expert Dr Anthony Fauci said if distribution goes well and enough Americans agree to get vaccinated, herd immunity could be achieved by the end of the northern summer. "If we do it efficiently enough by the second quarter of 2021, and by the end of summer or end of third quarter, we may actually have enough herd immunity protecting our society," he said. "And as we get to the end of 2021, we could approach very much some degree of normality that is close to where we were before." Robert Redfield, the director of the US Centres for Disease Control and Prevention, said on Friday (AEDT): "Probably for the next 60 to 90 days, we're going to have more deaths per day [from COVID-19] than we had on 9/11 or we had at Pearl Harbour." President-elect Joe Biden has expressed concern that the US will quickly run out of vaccines after the initial phase of distribution because the Trump administration passed on an opportunity to buy tens of millions of extra doses of Pfizer's vaccine. The European Medicines Agency (EMA) is due to complete its review of the Pfizer vaccine by December 29 and India is accelerating its review. Britain's AstraZeneca is seeking approval for its vaccine in Britain after announcing interim late-stage trial data on November 23. It had an average efficacy rate of 70 per cent and as much as 90 percent for a subgroup of trial participants who got a half dose first, followed by a full dose. However, it is not clear how the regulator will deal with the different dosages in the efficacy data in its assessment. While India is conducting an accelerated review, it has asked for more data. AstraZeneca is also in discussions with Europe. As its vaccines only need to be stored at fridge temperatures, it's still considered the best solution for developing nations battling coronavirus. Johnson & Johnson plans to deliver trial data this year or early in 2021, teeing it up for US approval in February if its shot is effective. China has five separate vaccines in third-stage trials around the world, and the United Arab Emirates has already approved a Sinaopharm shot after local trials, claiming it is 86 per cent effective. Novavax is running a late-stage trial in Britain with data due in the first quarter of 2021. It expects to start a large-scale trial in the United States this month. France's Sanofi and Britain's GlaxoSmithKline, however, announced a setback on December 11 in their attempts to develop a vaccine. The drugmakers said it showed an insufficient immune response in older people in mid-stage trials and that they would start a new study in February. with Katina Curtis, Reuters www.theage.com.au/world/north-america/us-regulators-approve-pfizer-vaccine-immunisations-to-start-in-days-20201212-p56mvo.html |
|